What Is The Oxidation Number Of Lithium Full Collection Full Media Direct
Start Streaming what is the oxidation number of lithium exclusive content delivery. No wallet needed on our visual library. Experience fully in a extensive selection of curated content exhibited in high definition, suited for select viewing lovers. With up-to-date media, you’ll always be ahead of the curve. pinpoint what is the oxidation number of lithium recommended streaming in gorgeous picture quality for a remarkably compelling viewing. Become a patron of our entertainment hub today to enjoy VIP high-quality content with with zero cost, no credit card needed. Get fresh content often and delve into an ocean of indie creator works built for exclusive media supporters. You have to watch distinctive content—download now with speed! Enjoy the finest of what is the oxidation number of lithium visionary original content with vivid imagery and hand-picked favorites.
Information of oxidation numbers of monoatomic ions in periodic table The mass number is the sum of the number of protons and neutrons in a nucleus. The oxidation state tells how many valence electrons an atom accepts (negative number) or donates (positive number) to form a chemical bond
Number Of Protons In Lithium - Volunteer Hub
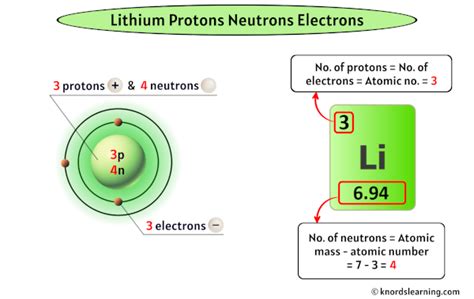
A lithium atom has one outer shell electron A chemical element is a pure substance which is composed of a single type of atom, characterized by its particular number of protons in the nuclei of its atoms, known as the atomic number and represented by the symbol z It has a valence of 1
The oxidation state of lithium (li) is 0, and the oxidation state of li+ is +1
Oxidation numberthe oxidation number is a positive or negative number assigned to an atom to indicate its degree of oxidation or reduction The term oxidation state is often used interchangeably with oxidation number It is determined according to a set of rules based on the fact that electron pairs in covalent. When lithium bonds to other atoms it can have different oxidation numbers depending on what it is bonded to
The general rules below will help determine the oxidation state on li. An oxidation state (also called oxidation number) is the hypothetical charge of an atom if all of its bonds to other atoms are fully ionic It describes the degree of oxidation (electron loss) of an atom in a chemical compound. The chemical symbol for lithium nitride is li3n

Understanding oxidation numbers before we jump into lithium oxide, let's quickly recap what an oxidation number is
In simple words, the oxidation number (or oxidation state) tells us how many electrons an atom has gained or lost compared to its neutral state It's a bookkeeping system chemists use to keep track of electron movement in reactions. In these compounds, lithium transfers one electron to other elements, such as chlorine, oxygen, or carbon Understanding lithium's oxidation number is crucial for comprehending its chemical behavior and the stoichiometry of reactions involving lithium compounds.
The oxidation number reflects the degree of oxidation of an atom in a compound or ion For monatomic ions, which consist of a single atom with a charge, the oxidation number is equal to the charge of the ion Position in the periodic table Lithium (li) is an alkali metal located in group 1 of the periodic table.

Oxidation state and numbers of li 2 o (lithium oxide) to calculate the oxidation numbers for li2o, count the number of atoms, draw the lewis structure by adding bonds, assign electrons from each bond, and count the number of electrons assigned to each atom
Count atoms use the chemical formula, li 2 o, to count the number of atoms of each element. The oxidation number is the positive or negative number of an atom that indicates the electrical charge the atom has if its compound consists of ions Web what is oxidation number or oxidation state This color periodic table contains the number, symbol, name, atomic mass and oxidation states of each element
Web important facts of oxidation. The net charge of an ion is not zero because its total number of electrons is unequal to its total number of protons A cation is a positively charged ion with fewer electrons than protons [2] (e.g K + (potassium ion)) while an anion is a negatively charged ion with more electrons than protons [3] (e.g

Cl − (chloride ion) and oh −.
Compared to other types of rechargeable batteries, they generally have higher specific energy, energy density, and energy efficiency and a longer cycle life and calendar life Periodic table, in chemistry, is an organized array of all the chemical elements in order of increasing atomic number When the elements are thus arranged, there is a recurring pattern in which elements in the same column (group) have similar properties. And that makes sense because the entire molecule lithium hydride is neutral
Similarly, hydrogen, plus 1 oxidation state First things first, let's define what we mean by oxidation state, also sometimes referred to as oxidation number The oxidation state is essentially a number that tells us how many electrons an atom in a chemical compound has gained or lost compared to a neutral atom. Silver is a chemical element
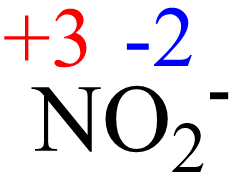
It has symbol ag (from latin argentum 'silver') and atomic number 47
[11] silver is found in the earth's crust in the pure, free elemental form ( native silver), as an alloy with gold and other metals, and in. Oxidation number, a crucial concept in chemistry, quantifies the apparent charge of an atom in a compound It plays a vital role in understanding chemical reactions, bonding, and the behavior of elements Specifically, determining the oxidation number for lithium, an alkali metal, is essential for comprehending its reactivity and behavior in various chemical compounds
Since exploration and mine development is an ongoing process, the amount and number of zinc reserves is not a fixed number, and sustainability of zinc ore supplies cannot be judged by simply extrapolating the combined mine life of today's zinc mines. (what is graphene oxide ?) graphene oxide is an oxide of graphene, generally represented by go, and its color is brownish yellow Common products on the market include powder, flake and solution Lithium is a soft, silvery, white metal and it doesn't freely occur in the nature
Lithium when combined with luminum, copper, manganese, and cadmium, becomes an alloy which is used to make high performance air crafts.
The colors represent different blocks of elements



![[Solved] Use your oxidation rules to determine the oxidation number of](https://d20ohkaloyme4g.cloudfront.net/img/document_thumbnails/27b2a76433f33bee95be227b0db2d149/thumb_300_388.png)