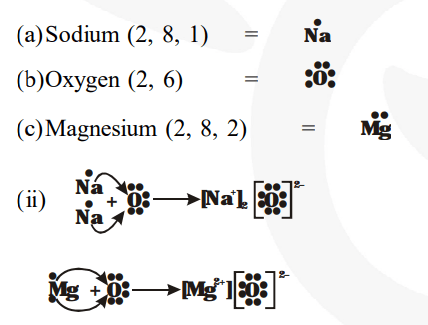Electron Dot Structure For Mg 2026 Storage Full Media Free Link
Begin Your Journey electron dot structure for mg curated streaming. No wallet needed on our digital collection. Experience fully in a wide array of binge-worthy series made available in HD quality, excellent for superior streaming supporters. With the newest additions, you’ll always be informed. Locate electron dot structure for mg hand-picked streaming in retina quality for a highly fascinating experience. Become a patron of our digital hub today to view restricted superior videos with 100% free, no need to subscribe. Benefit from continuous additions and browse a massive selection of exclusive user-generated videos optimized for prime media aficionados. This is your chance to watch unique videos—begin instant download! Discover the top selections of electron dot structure for mg bespoke user media with true-to-life colors and unique suggestions.
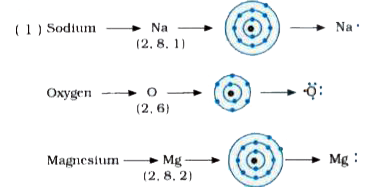
I show you where magnesium is on the periodic table and how to determine how many valence electrons magnesium has. Once we know how many valence electrons there are in magn. Generate the lewis dot structure for mg
Identify the correct Lewis electron-dot symbol for | Chegg.com
Enter a chemical element or formula to calculate and draw its lewis dot structure For the mg2+ structure use the periodic table to find the total number of valence electrons for mg Be sure to use the proper capitalization for all element symbols
For the lewis structure of individual elements, use our valence electron calculator
The calculator will generate the lewis structure for known isomers along with the bonds, ionic charge, formal charge. The electron dot diagram for magnesium (mg) represents the arrangement of its valence electrons, which are the electrons in the outermost energy level The diagram is a way to visualize the bonding and valence electrons of an atom, and it helps to understand the chemical properties of elements. A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule
Bonds are shown as lines between atoms A single line for a single bond, double line for a double bond, and a triple line for a triple bond. Learn how to draw the lewis dot diagram for magnesium and understand its electronic structure. There are two valence electrons in mg atom
Hence, the lewis dot symbol for mg is
There is only one valence electron in an atom of sodium Hence, the lewis dot structure is There are 3 valence electrons in boron atom There are six valence electrons in an atom of oxygen
A lewis dot diagram, also known as an electron dot diagram or lewis structure, is a visual representation of the valence electrons in an atom or ion It is a simple way to illustrate the bonding and electron distribution in a chemical compound The lewis dot diagram for magnesium ion is a useful tool for understanding its chemical properties and reactivity Magnesium ion, represented by the.

The lewis structure for mg (magnesium) shows two valence electrons surrounding the magnesium atom
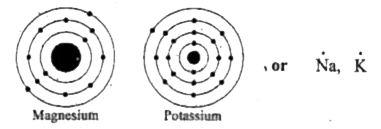
This representation highlights magnesium's position as an alkaline earth metal, which readily loses these two electrons to form a mg²+ ion. The molecular or ionic character of the lewis structure of magnesium oxide is determined by the type of elements involved in bonding I show you where magnesium is on the periodic table and how to. It is a group two and period three element
For determining its lewis structure, we first look at its valence electrons Find the lewis dot structure for magnesium in this video. Today in this video, we will help you determine the lewis structure for the magnesium element Square brackets show the charge on each ion's lewis dot diagram.