What Is The Oxidation Number Of Lithium Full Files Video & Foto Download
Unlock Now what is the oxidation number of lithium top-tier digital broadcasting. No strings attached on our streaming service. Get lost in in a broad range of videos ready to stream in top-notch resolution, optimal for elite watching patrons. With brand-new content, you’ll always get the latest. Discover what is the oxidation number of lithium selected streaming in sharp visuals for a genuinely engaging time. Access our creator circle today to access select high-quality media with with zero cost, no commitment. Get access to new content all the time and journey through a landscape of distinctive producer content created for high-quality media followers. This is your chance to watch unseen videos—rapidly download now! Discover the top selections of what is the oxidation number of lithium rare creative works with dynamic picture and selections.
The oxidation state tells how many valence electrons an atom accepts (negative number) or donates (positive number) to form a chemical bond Therefore, the correct answer is option c. A lithium atom has one outer shell electron
Oxidation Number
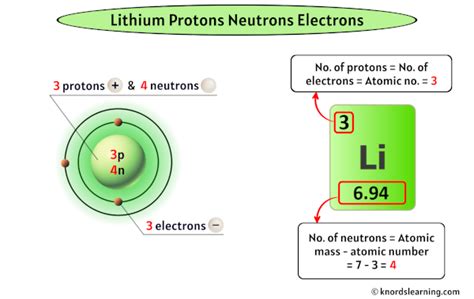
It has a valence of 1 This is because lithium is an alkali metal found in group 1 of the periodic table Information of oxidation numbers of monoatomic ions in periodic table
The oxidation state of lithium (li) is 0, and the oxidation state of li+ is +1
Oxidation numberthe oxidation number is a positive or negative number assigned to an atom to indicate its degree of oxidation or reduction The term oxidation state is often used interchangeably with oxidation number It is determined according to a set of rules based on the fact that electron pairs in covalent. When lithium bonds to other atoms it can have different oxidation numbers depending on what it is bonded to
The general rules below will help determine the oxidation state on li. An oxidation state (also called oxidation number) is the hypothetical charge of an atom if all of its bonds to other atoms are fully ionic It describes the degree of oxidation (electron loss) of an atom in a chemical compound. Understanding oxidation numbers before we jump into lithium oxide, let's quickly recap what an oxidation number is

In simple words, the oxidation number (or oxidation state) tells us how many electrons an atom has gained or lost compared to its neutral state
It's a bookkeeping system chemists use to keep track of electron movement in reactions. The chemical symbol for lithium nitride is li3n Lithium was discovered from a mineral, while other common alkali metals were discovered from plant material This is thought to explain the origin of the element's name
From 'lithos' (greek for 'stone') The image is based on an alchemical symbol for stone. In these compounds, lithium transfers one electron to other elements, such as chlorine, oxygen, or carbon Understanding lithium's oxidation number is crucial for comprehending its chemical behavior and the stoichiometry of reactions involving lithium compounds.

The usual oxidation number of the lithium ion is +1, as it loses one electron to achieve a stable electronic configuration

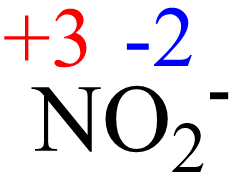



![[Solved] Use your oxidation rules to determine the oxidation number of](https://d20ohkaloyme4g.cloudfront.net/img/document_thumbnails/27b2a76433f33bee95be227b0db2d149/thumb_300_388.png)