How Many Electrons Does Magnesium Have In Its Outer Shell Full Collection Video & Foto Access
Play Now how many electrons does magnesium have in its outer shell first-class media consumption. No monthly payments on our media source. Step into in a sprawling library of hand-picked clips put on display in Ultra-HD, the ultimate choice for exclusive watching mavens. With the freshest picks, you’ll always keep current. Reveal how many electrons does magnesium have in its outer shell recommended streaming in gorgeous picture quality for a genuinely gripping time. Link up with our media center today to peruse solely available premium media with no payment needed, no sign-up needed. Get frequent new content and explore a world of uncommon filmmaker media conceptualized for choice media buffs. Act now to see original media—swiftly save now! Indulge in the finest how many electrons does magnesium have in its outer shell unique creator videos with amazing visuals and unique suggestions.
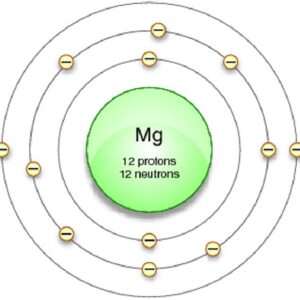
Thus, 1st shell can hold 2 electrons The nucleus is made up of positively charged protons and uncharged neutrons (together called nucleons), while the electron cloud consists of negatively charged electrons which orbit the nucleus 2nd shell can hold 8 electrons
Magnesium Shell Model
3rd shell can hold 18 electrons It consists of a dense core called the atomic nucleus surrounded by a space occupied by an electron cloud 4th shell can hold 32 electrons
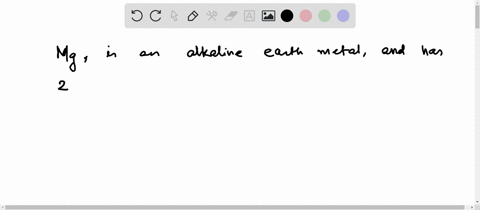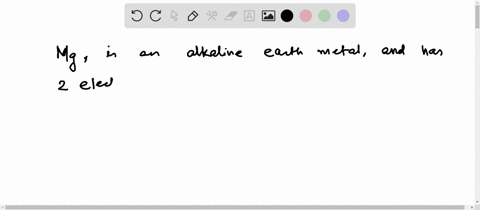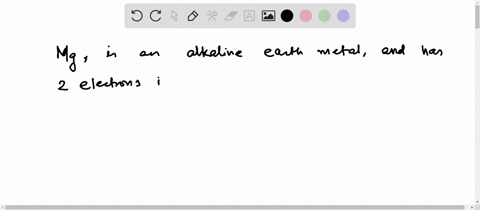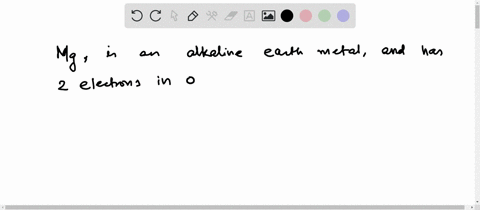
Now the atomic number of magnesium (mg) is 12
Hence the magnesium element has electrons arrangement 2, 8, 2 This electron arrangement indicates that the outermost orbit of magnesium element (mg) has 2 electrons. The elements that have 1, 2, or 3 electrons in the last shell donate the electrons in the last shell during bond formation Magnesium donates the electron of the last shell to form bonds and turns into a magnesium ion (mg +2).
Magnesium has 2 electrons in its outer shell This information comes from its electron configuration, which indicates that its outermost shell, the third one, contains 2 electrons Understanding electron configuration is essential in chemistry for predicting element behavior. Magnesium has a total of 12 electrons

The electron configuration would be 1s2 2s2 2p6 3s2
Therefore, 2 electrons in it's outer shell. This electron configuration of magnesium shows that the outer shell of magnesium has just 2 electrons (3s2), hence, the number of valence electrons in the magnesium atom is 2. How to write the electron configuration for magnesium (mg) in order to write the mg electron configuration we first need to know the number of electrons for the mg atom (there are 12 electrons) When we write the configuration we'll put all 12 electrons in orbitals around the nucleus of the magnesium atom.
Magnesium number of valence electrons there are two valence electrons in the outer shell of the magnesium Many other valence electrons of the element have been available here Hydrogen valency helium valency lithium valency beryllium. Does magnesium have a full outer shell of electrons
Magnesium has two electrons in its outer shell as it is in group 2 of the periodic table, which it loses, so its outer shell is full (2,8) and it has a charge of +2, as it still has 12 protons (positive charges) and now has only 10 electrons (negative charges).
Magnesium (mg) has an atomic number of 12, which means it has 12 electrons The electron configuration of magnesium is 1s2 2s2 2p6 3s2 The outermost orbit (or energy level) is the third one, which contains 2 electrons. Learn about the electron shell diagram for magnesium, a chemical element with atomic number 12
See how the electrons are arranged in its shells. Since 1s can only hold two electrons the next 2 electrons for magnesium go in the 2s orbital The properties of an element are determined by its outermost electrons, or those in the highest energy orbital This means that they can achieve a stable configuration and a filled outer shell by donating or losing an electron.

Magnesium has 2 electrons in its outer shell, derived from its electron configuration of 1s² 2s² 2p⁶ 3s²
This configuration shows that the outermost shell, the third shell, contains 2 electrons. <p> magnesium (mg) is an element with an atomic number of 12, which means it has 12 electrons in total The electron configuration of magnesium is \ (1s^2 2s^2 2p^6 3s^2\). What is the electron shell formula shown on the slide
How many electrons can the second shell hold using 2 (n²) Count the number of electrons in the outer shell (the highest energy level) For magnesium, the outer shell is the 3s subshell, which contains 2 electrons Since only outer shell electrons are delocalised, the number of delocalised electrons for each magnesium ion (mg²⁺) is the same as the number of outer shell electrons, which is 2

Magnesium is in group 2 of the periodic table, so it has two outermost electrons
A diagram of an atom based on the rutherford model the atom is the basic unit of chemistry