Mercury Number Of Protons Neutrons And Electrons Digital Vault Vids & Pics Free Link
Get Started mercury number of protons neutrons and electrons elite viewing. Pay-free subscription on our media destination. Lose yourself in a wide array of curated content available in cinema-grade picture, the ultimate choice for high-quality streaming enthusiasts. With current media, you’ll always stay on top of. Watch mercury number of protons neutrons and electrons personalized streaming in ultra-HD clarity for a highly fascinating experience. Access our content portal today to watch restricted superior videos with free of charge, no commitment. Be happy with constant refreshments and journey through a landscape of distinctive producer content produced for premium media buffs. Be sure not to miss uncommon recordings—download fast now! Get the premium experience of mercury number of protons neutrons and electrons visionary original content with amazing visuals and featured choices.
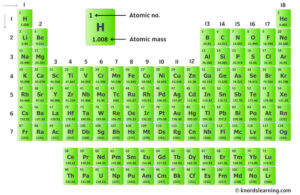
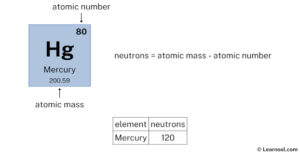
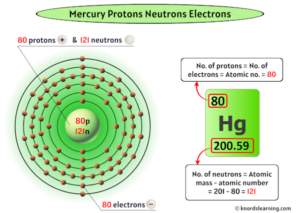
It consists of 80 protons, 120 neutrons, and 80 electrons. During the years from 1930 to 1945, a wide variety of investigations confirmed that the primary cosmic rays are mostly protons, and the secondary radiation produced in the atmosphere is primarily electrons, photons and muons. Mercury is the 80th element in the periodic table and has a symbol of hg and atomic number of 80
Mercury Protons Neutrons Electrons (And How to Find them?)
It has an atomic weight of 200.592 and a mass number of 202 Step 1 of 3the atom can be represented as, step 2 of 3from the above figure, the number of protons of an atom is 29 so the atomic number of the atom is 29.and the number of It is located in group twelve, period six and block d of the periodic table
Heavy silvery liquid metallic element.
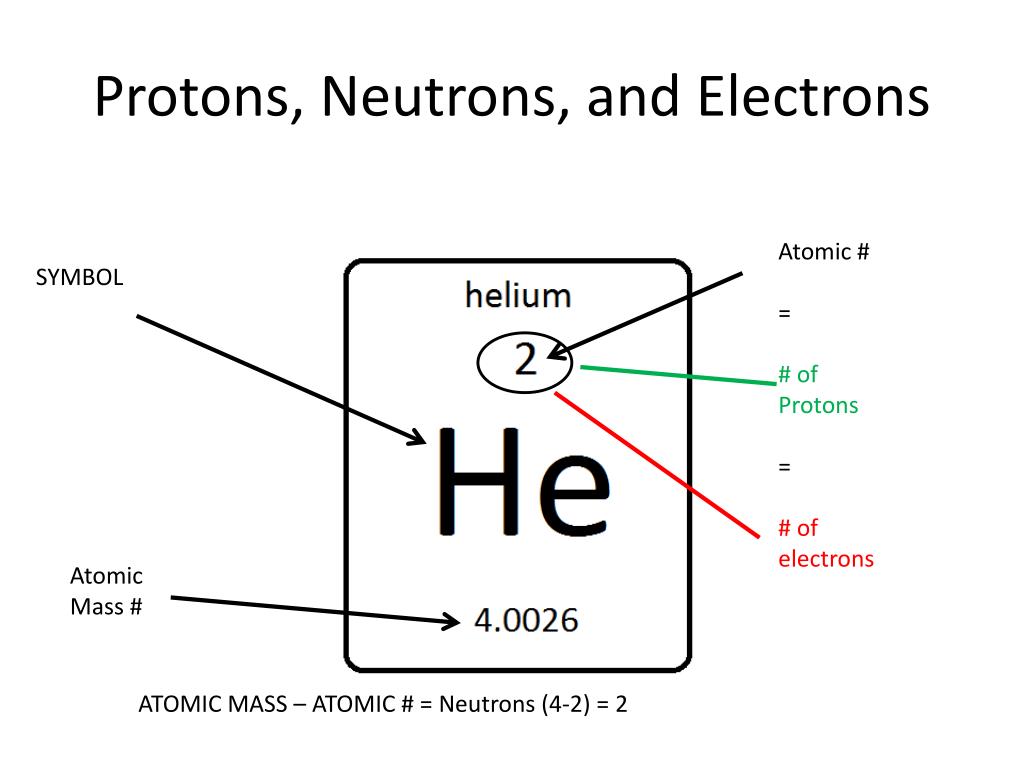
The atomic number of an element is equal to the number of protons and electrons in that element Therefore, a mercury atom has eighty protons and eighty electrons The number of neutrons in an atom can be determined by the difference between the atomic mass and the number of protons. Learn how to find the number of protons, neutrons and electrons in mercury atom using simple methods
The web page provides the atomic number, mass and number of each particle for mercury (hg). All atomic nuclei of the chemical element mercury are summarized under mercury isotopes These all consist of an atomic nucleus with 80 protons and, in the uncharged state, 80 electrons The difference between each isotope of mercury is based on the number of neutrons in the nucleus
Naturally occurring mercury isotopes the natural mercury deposits on earth consist of a mix of seven different.
Basic information | atomic structure | isotopes | related links | citing this page basic information name 356.58 °c (629.73 k, 673.844 °f) number of protons/electrons The atomic number of mercury (hg) is 80 Since atomic number of an atom= no
Of protons in mercury is 80. Atomic number & mass number the atomic number (80) represents the number of protons in the nucleus The mass number (201) is the sum of protons and neutrons in the nucleus. A complete understanding of mercury's atomic structure integrates the roles of its 80 protons, its typically 80 electrons (in a neutral atom), and its varying number of neutrons.

Photo courtesy of about.com chemical elements jefferson labs web elements wikipedia
29 protons, 29 neutrons, 27 electrons 29 protons, 35 neutrons, 27 electrons 31 protons, 35 neutrons, 29 electrons 31 protons, 33 neutrons, 29 electrons The atomic number indicates the number of protons in an atom, defining the element, while atomic mass is the sum of protons and neutrons These values are calculated based on the element's isotopes and their relative abundances Noble gases are characterized by their lack of reactivity due to having a full valence shell, making them stable.
The mass number is equal to a) the sum of the number of protons and neutrons B) the sum of the number of the neutrons and electrons C) the sum of the number of protons, neutrons, and electrons D) the sum of the number of the electrons and protons

A chem 161 combustion analysis of an unknown compound containing only carbon and.
Study with quizlet and memorize flashcards containing terms like identify the element that has an atomic number of 15., an atom of ¹³¹xe contains ________ electrons., how many electrons are in nickel Made up of protons and neutrons (nucleons) electrons orbit the nucleus in defined energy levels and distances electrical charge is.a characteristic of matter if an atom has an equal number of protons and electrons. Atoms of the same element with different numbers of neutrons How do you determine the number of protons, neutrons, and electrons from an atomic symbol
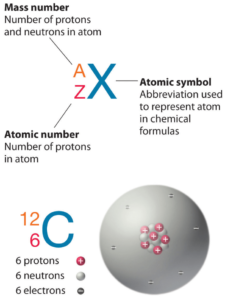
By analyzing the atomic symbol which includes the atomic number and mass number What is an electron configuration A representation of the arrangement of electrons in an atom. Therefore, the atomic number also indicates the number of electrons in an atom
The total number of protons and neutrons in an atom is called its mass number (a)
The number of neutrons is therefore the difference between the mass number and the atomic number The number of neutrons can be calculated using the formula Jove atomic structure (proton, neutron and electron) all matter is made up of tiny particles called atoms Each atom has three primary subatomic particles
Give the number of protons, neutrons, and electrons for ²nnnal What is the symbol and charge of an ion with 26 protons, 30 neutrons, and 23 electrons Convert 22 °c to kelvin 10b = 10 amu (19%) 11b = 11 amu (80%) calculate the average atomic mass.

The booklet must contain the following
Students will be required to find the number of protons, electrons and neutrons of given elements Students are to have to neatly draw the planetary model for each along with the electron configuration of each. The density of air at room temperature and at sea level is 1.29 g/l What is its density in units of g/ml
0.00129 g/ml 300 list the number of protons, neutrons, and electrons for 32 ca 2+ protons A particular atom contains 29 electrons, 34 neutrons, and 29 protons What is the identity of this element, and what is its atomic number