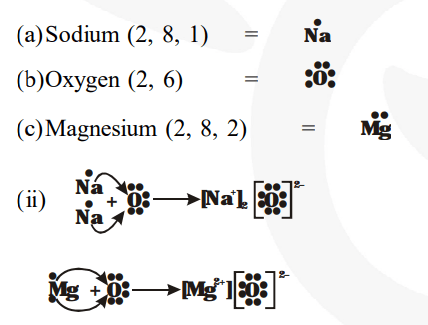Electron Dot Structure For Mg 2026 Vault Media Files Fast Access
Begin Your Journey electron dot structure for mg hand-selected online video. No strings attached on our digital playhouse. Get captivated by in a sprawling library of selections available in superior quality, optimal for exclusive viewing connoisseurs. With up-to-date media, you’ll always have the latest info. Reveal electron dot structure for mg preferred streaming in photorealistic detail for a truly enthralling experience. Sign up today with our video library today to see unique top-tier videos with at no cost, registration not required. Receive consistent updates and navigate a world of one-of-a-kind creator videos engineered for exclusive media followers. Seize the opportunity for specialist clips—download fast now! Explore the pinnacle of electron dot structure for mg rare creative works with exquisite resolution and preferred content.
Generate the lewis dot structure for mg Find the lewis dot structure for magnesium in this video. Enter a chemical element or formula to calculate and draw its lewis dot structure
Lewis Electron Dot Structure Molecule Hydrogen Stock Vector (Royalty
Be sure to use the proper capitalization for all element symbols For determining its lewis structure, we first look at its valence electrons For the lewis structure of individual elements, use our valence electron calculator
The calculator will generate the lewis structure for known isomers along with the bonds, ionic charge, formal charge.
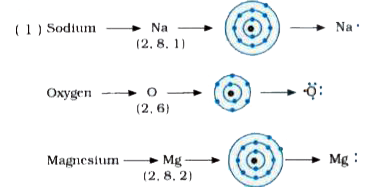
I show you where magnesium is on the periodic table and how to determine how many valence electrons magnesium has. A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule Bonds are shown as lines between atoms A single line for a single bond, double line for a double bond, and a triple line for a triple bond.
Learn how to create an electron dot diagram for magnesium (mg) and understand its electron configuration and bonding. There are two valence electrons in mg atom Hence, the lewis dot symbol for mg is There is only one valence electron in an atom of sodium
Hence, the lewis dot structure is
There are 3 valence electrons in boron atom There are six valence electrons in an atom of oxygen Learn how to draw the lewis dot diagram for magnesium and understand its electronic structure. Write the electron dot structure for magnesium and chlorine
Show the formation of magnesium chloride by the transfer of electrons What are the ions present in this compound? The lewis dot diagram, also known as the electron dot diagram or lewis structure, is a graphical representation of the valence electrons in an atom or molecule It was introduced by gilbert n

Lewis in 1916 as a way to visualize the bonding between atoms and predict molecular geometry
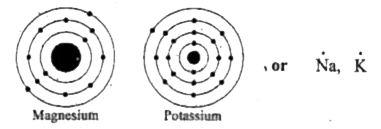
Magnesium (mg) is a chemical element with the symbol mg and atomic number 12 It is an alkaline earth metal. The lewis structure for mg (magnesium) shows two valence electrons surrounding the magnesium atom This representation highlights magnesium's position as an alkaline earth metal, which readily loses these two electrons to form a mg²+ ion.
It is a group two and period three element