How Many Electrons Does Magnesium Have In Its Outer Shell 2026 Vault HD Media Download
Begin Immediately how many electrons does magnesium have in its outer shell prime streaming. No strings attached on our content platform. Become absorbed in in a massive assortment of clips exhibited in flawless visuals, great for dedicated viewing connoisseurs. With fresh content, you’ll always stay in the loop. Uncover how many electrons does magnesium have in its outer shell expertly chosen streaming in impressive definition for a genuinely gripping time. Sign up today with our media center today to check out special deluxe content with no payment needed, free to access. Look forward to constant updates and browse a massive selection of uncommon filmmaker media crafted for superior media supporters. Don't forget to get unique videos—click for instant download! Witness the ultimate how many electrons does magnesium have in its outer shell distinctive producer content with flawless imaging and editor's choices.
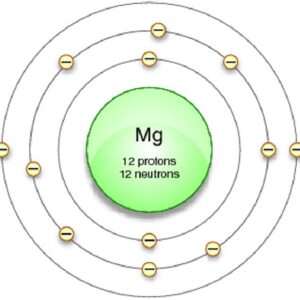
Thus, 1st shell can hold 2 electrons Each stationary orbit or shell is associated with a definite amount of energy. 2nd shell can hold 8 electrons
PPT - How many valence electrons does magnesium have? PowerPoint
3rd shell can hold 18 electrons Shells have stationary energy levels, the energy of each shell is constant 4th shell can hold 32 electrons
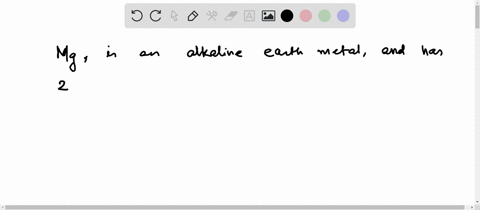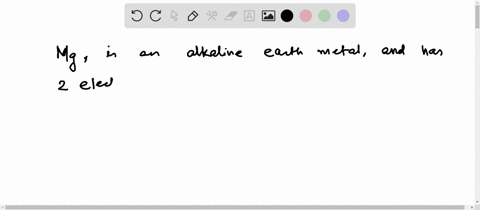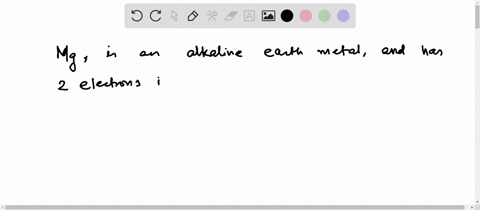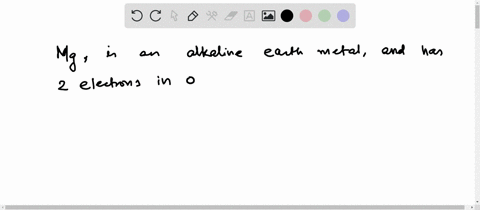
Now the atomic number of magnesium (mg) is 12
Hence the magnesium element has electrons arrangement 2, 8, 2 This electron arrangement indicates that the outermost orbit of magnesium element (mg) has 2 electrons. The elements that have 1, 2, or 3 electrons in the last shell donate the electrons in the last shell during bond formation Magnesium donates the electron of the last shell to form bonds and turns into a magnesium ion (mg +2).
Magnesium has 2 electrons in its outer shell This information comes from its electron configuration, which indicates that its outermost shell, the third one, contains 2 electrons Understanding electron configuration is essential in chemistry for predicting element behavior. This electron configuration of magnesium shows that the outer shell of magnesium has just 2 electrons (3s2), hence, the number of valence electrons in the magnesium atom is 2.

Magnesium has a total of 12 electrons
The electron configuration would be 1s2 2s2 2p6 3s2 Therefore, 2 electrons in it's outer shell. How to write the electron configuration for magnesium (mg) in order to write the mg electron configuration we first need to know the number of electrons for the mg atom (there are 12 electrons) When we write the configuration we'll put all 12 electrons in orbitals around the nucleus of the magnesium atom.
From wikipedia, the free encyclopedia magnesium is a chemical element with symbol mg and atomic number 12 It is a shiny gray solid which bears a close physical resemblance to the other five elements in the second column (group 2, or alkaline earth metals) of the periodic table:they each have the same electron configuration in their outer electron shell producing a similar crystal structure. Does magnesium have a full outer shell of electrons Magnesium has two electrons in its outer shell as it is in group 2 of the periodic table, which it loses, so its outer shell is full (2,8) and it has a charge of +2, as it still has 12 protons (positive charges) and now has only 10 electrons (negative charges).
Magnesium number of valence electrons there are two valence electrons in the outer shell of the magnesium
Many other valence electrons of the element have been available here Hydrogen valency helium valency lithium valency beryllium. A diagram of an atom based on the rutherford model the atom is the basic unit of chemistry It consists of a dense core called the atomic nucleus surrounded by a space occupied by an electron cloud
The nucleus is made up of positively charged protons and uncharged neutrons (together called nucleons), while the electron cloud consists of negatively charged electrons which orbit the nucleus Hydrogen with 1 proton, 0 neutrons, and 1 electron isotope atoms that have the same number of protoon and electrons but vary in the number neutrons isotopes have the same ______ but different _________ atomic number, atomic masses isotopes all behave identically to one another in_______ How are electrons arranged in elements with atomic numbers 1 through 10 Based on its atomic number, how many electrons does a hydrogen atom have

The valence shell of an element with configuration 1s²2s²2p⁶3s²3p³ contains
Outer shell is 3 → electrons = 3s²3p³ = 5. Iodine has an electron configuration of [kr]5s 2 4d 10 5p 5, with the seven electrons in the fifth and outermost shell being its valence electrons. Valence electro n valence electron refers to the number of electrons on the outer shell of an atom. The word supernova was coined by walter baade and fritz zwicky, who began using it in astrophysics lectures in 1931
[1][2] its first use in a journal article came the following year in a publication by knut lundmark, who may have coined it independently Atoms tend to gain, lose, or share electrons to achieve a full outer shell of eight electrons, leading to stable configurations Involves a 'sea of electrons' that allows metals to conduct electricity and exhibit malleability and ductility. A sodium atom and a sodium ion have the same a number of neutronsb number of electrons c electric charge d electronic structure

The first one has 2 electrons in the inner shell and 6 electrons in the outer shell
The second one has 2 electrons in the inner shell and 6 electrons in the outer shell. Magnesium, located in column 2 (or group 2) has 2 electrons in its valence shell It will give up 2 electrons to have the electron configuration of neon, as opposed to gaining 6 electrons. For (a) magnesium, a member of group 2 (alkaline earth metals), there are 2 electrons in its outer shell
(b) cobalt, being a transition metal, it's outer shell is the 4s subshell which has 2 electrons. According to bohr's atomic model electrons revolve around the nucleus in a specific circular path known as orbit or called a shell