How Many Electrons Are In A Neutral Barium Atom 2026 Archive Vids & Images Instant
Get Started how many electrons are in a neutral barium atom choice digital broadcasting. Free from subscriptions on our entertainment center. Step into in a wide array of curated content unveiled in top-notch resolution, a must-have for passionate viewing viewers. With the newest additions, you’ll always stay in the loop. Check out how many electrons are in a neutral barium atom expertly chosen streaming in crystal-clear visuals for a sensory delight. Enroll in our content collection today to get access to solely available premium media with absolutely no cost to you, no need to subscribe. Get frequent new content and explore a world of singular artist creations designed for select media followers. Don't forget to get rare footage—click for instant download! Access the best of how many electrons are in a neutral barium atom specialized creator content with dynamic picture and curated lists.
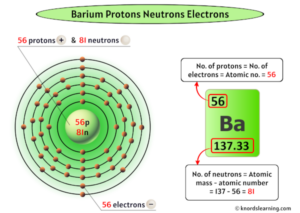
Barium is the 56th element of the periodic table How many unpaired electrons are present in fe2+? Barium atoms have 56 electrons and the shell structure is 2.8.18.18.8.2
Barium protons neutrons electrons - Learnool
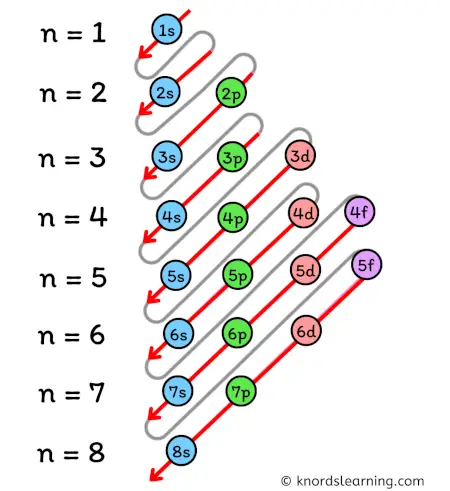
The ground state electron configuration of ground state gaseous neutral barium is [xe] Hence, the number of unpaired electron in n2+ is 1 6s2 and the term symbol is 1s0.
A neutral fluorine atom (f) has atomic number 9
How many electrons does it contain These elements typically lose their two valence electrons to achieve a stable electron configuration, forming ions with a 2+ charge (ba²⁺) In summary, the neutral barium atom contains 56 total electrons and 2 valence electrons, with the latter located in the outermost 6s orbital. A neutral barium atom has 56 electrons
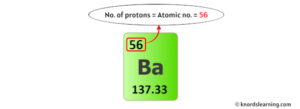
This is because the atomic number of barium is 56, which corresponds to the number of electrons it has when it is electrically neutral. A single atom of barium has 56 protons in its nucelus and in its elemental form has 56 electrons in electron orbitals surrounding the nucelus Most naturally ocurring barium atoms have 82 neutrons in their nucleus, but there are seven stable isotopes (different numbers of neutrons), naturally occuring. Explanation <p> barium (ba) has an atomic number of 56

For neutral atoms, the atomic number also denotes the number of electrons in an atom
Therefore, a neutral atom of barium will contain 56 electrons.</p> 94 click to rate: The atomic mass of barium is 137.27, so we'll take the roundup value as 137 And the atomic number of barium is 56 Subtract the atomic number (56) from the atomic mass (137)
Barium is the 56th element in the periodic table and the symbol is 'ba' Barium has an atomic number of 56, which means that its atom has 56 electrons around its nucleus The electron configuration of barium is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 10 5p 6 6s 2, which means that the first two electrons enter the 1s orbital. What neutral elements has the following electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d3
If you are referring to a neutral atom, then vanadium (v) has that particular electron configuration
How many unpaired electron are present in n2+