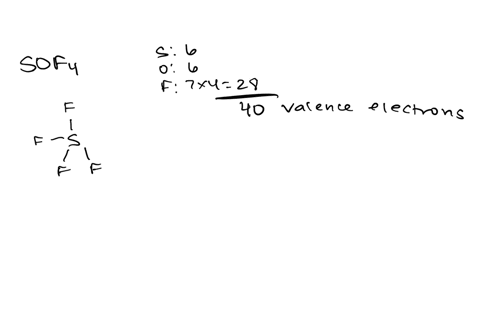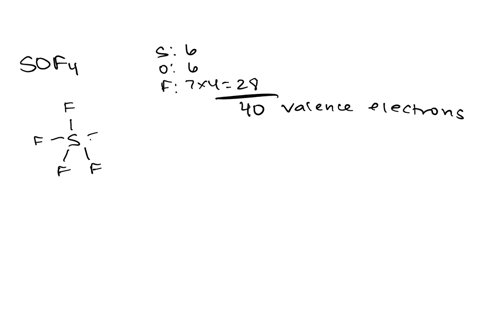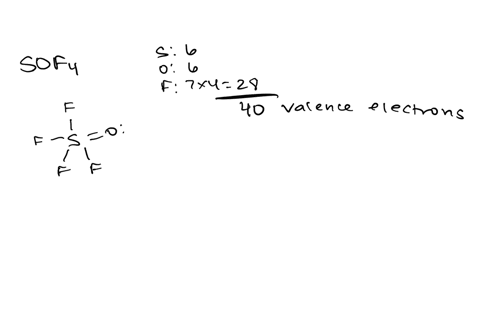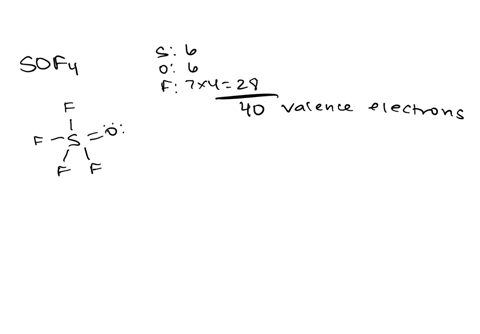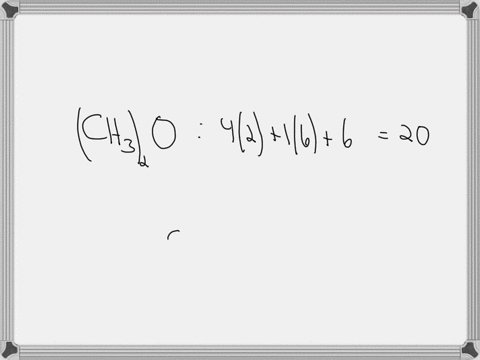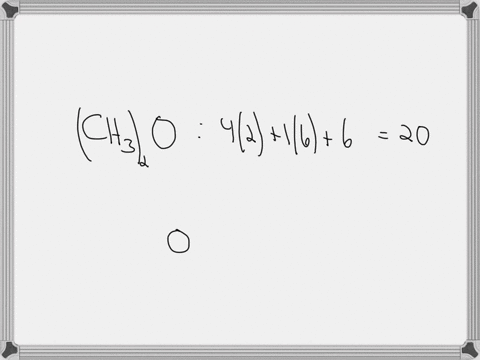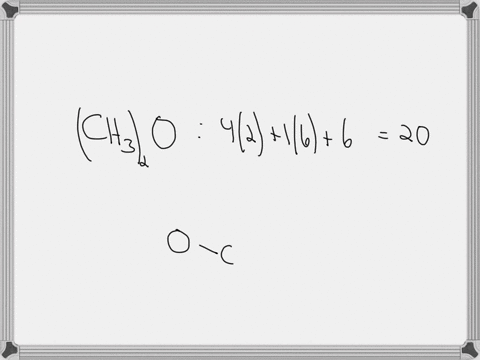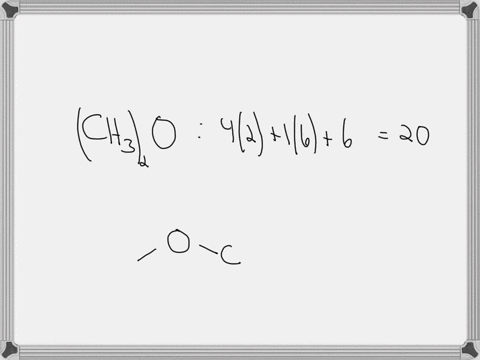Lewis Structure Of Cso Full Library Vids & Pics Full Link
Begin Immediately lewis structure of cso signature content delivery. Completely free on our streaming service. Become absorbed in in a ocean of videos of clips highlighted in HD quality, great for exclusive streaming fanatics. With newly added videos, you’ll always never miss a thing. pinpoint lewis structure of cso expertly chosen streaming in impressive definition for a sensory delight. Link up with our media world today to enjoy private first-class media with with zero cost, no credit card needed. Enjoy regular updates and explore a world of exclusive user-generated videos made for deluxe media addicts. You won't want to miss specialist clips—swiftly save now! Explore the pinnacle of lewis structure of cso unique creator videos with brilliant quality and unique suggestions.
The molecule polar or draw the lewis structure of cso and then determine nonpolar watch the full video at:.more This chemistry video provides a basic introduction into how to draw lewis structures of common molecules such as cl2, o2, of2, ch4, nh3, h2o, c2h2, and n2h4. Generate the lewis structure to see the valance electrons for a molecule or chemical element.
Cso Lewis Structure
This guide will provide a clear, visual explanation of how to draw the lewis structure for carbonyl sulfide (cso), a molecule with interesting properties Lewis structures show each atom and its position in the structure of the molecule using its chemical symbol. What is a lewis structure
Lewis structure generator creates chemical structure diagrams for compounds.
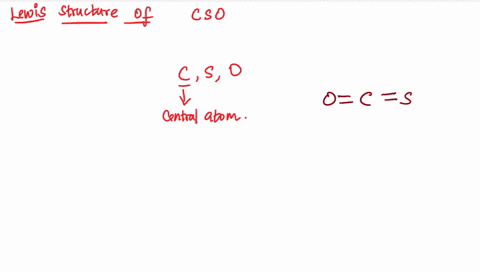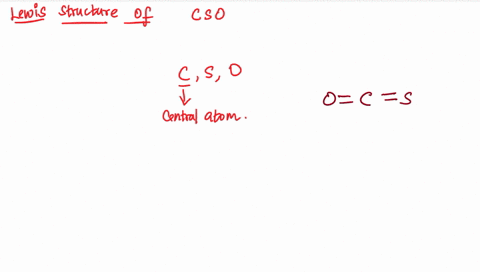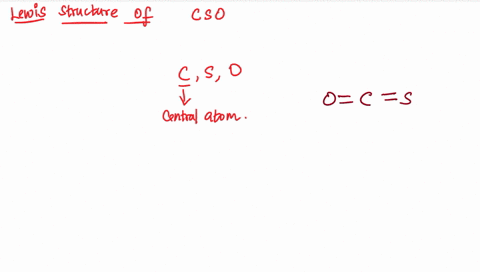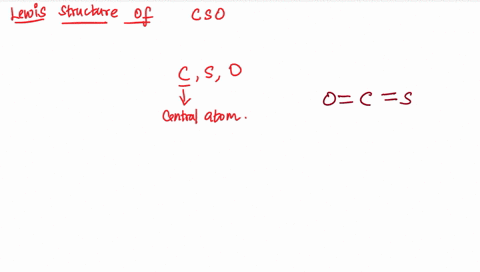
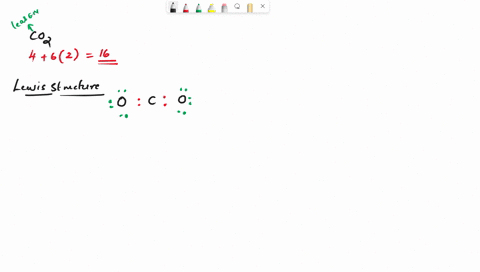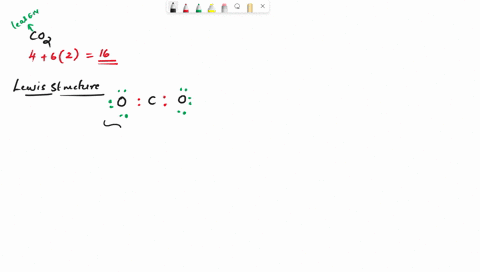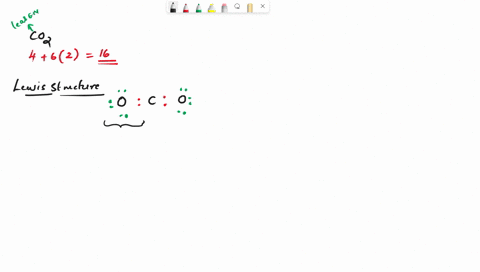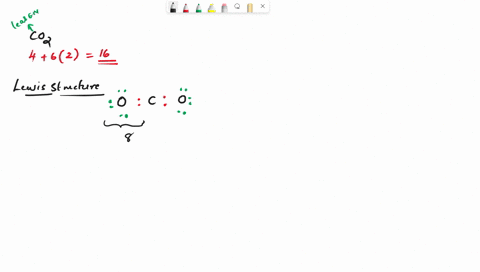
Draw the lewis structure of carbonyl sulfide (cso) by first counting the total valence electrons Carbon (c) has 4, sulfur (s) has 6, and oxygen (o) has 6, giving a total of 16 valence electrons. Draw the lewis structure of cso and then determine the molecules polar or nonpolar a) nonpolar + b) 1 answer below » 11 + users viewed 3 + downloaded solutions maryland, us mostly asked from draw the lewis structure of cso and then determine the molecules polar or nonpolar a) nonpolar + b) polar predict the product in the reaction shown below.
Draw the lewis structure of cso and then determine if the molecule is polar or nonpolar.a) nonpolarb) polar Draw the lewis structure of cso and then determine if the molecule is polar or nonpolar Square new structure a nonpolar b polar | question ai / questionoctober 11, 2024 Ö=c=s click to edit molecule a) nonpolar b) polar
This video discusses if sco or ocs is polar or nonpolar in addition to drawing the lewis structure of cso.
The lewis structure, a visual representation of the bonding and lone pairs of electrons in a molecule, provides valuable insights into these aspects The lewis structure for the molecule cso has c as the central atom and contains two single bonds two double bonds one double bond and one triple bond two triple bonds question 6 which atom is capable of having an incomplete octet? The lewis structure, proposed by gilbert newton lewis, who introduced it for the first time in 1916, is a graphic representation of the sharing of electrons that occurs in chemical bonds between atoms of the same or different species. Practice drawing these lewis structures and don't worry we will go over all the answers step by step
This video will explain how to find the formal charges. Solution for below is a lewis structure of cso Draw two resonance structures of this molecule Assign any formal charges if necessary

B) now look at your…
Lewis structure understanding the lewis structure is crucial when dealing with molecules like sulfine (h 2 cso) A lewis structure is a diagram that displays the bonds between atoms and the lone pairs of electrons in a molecule To draw the structure, arrange the atoms to reflect their electronegativities and bonding capabilities. In this video you'll learn how to draw lewis dot structures for covalent compounds
The video covers the basic lewis structures you'll see in an introductor. Lewis structures extend the concept of the electron dot diagram by adding lines between atoms to represent shared pairs in a chemical bond